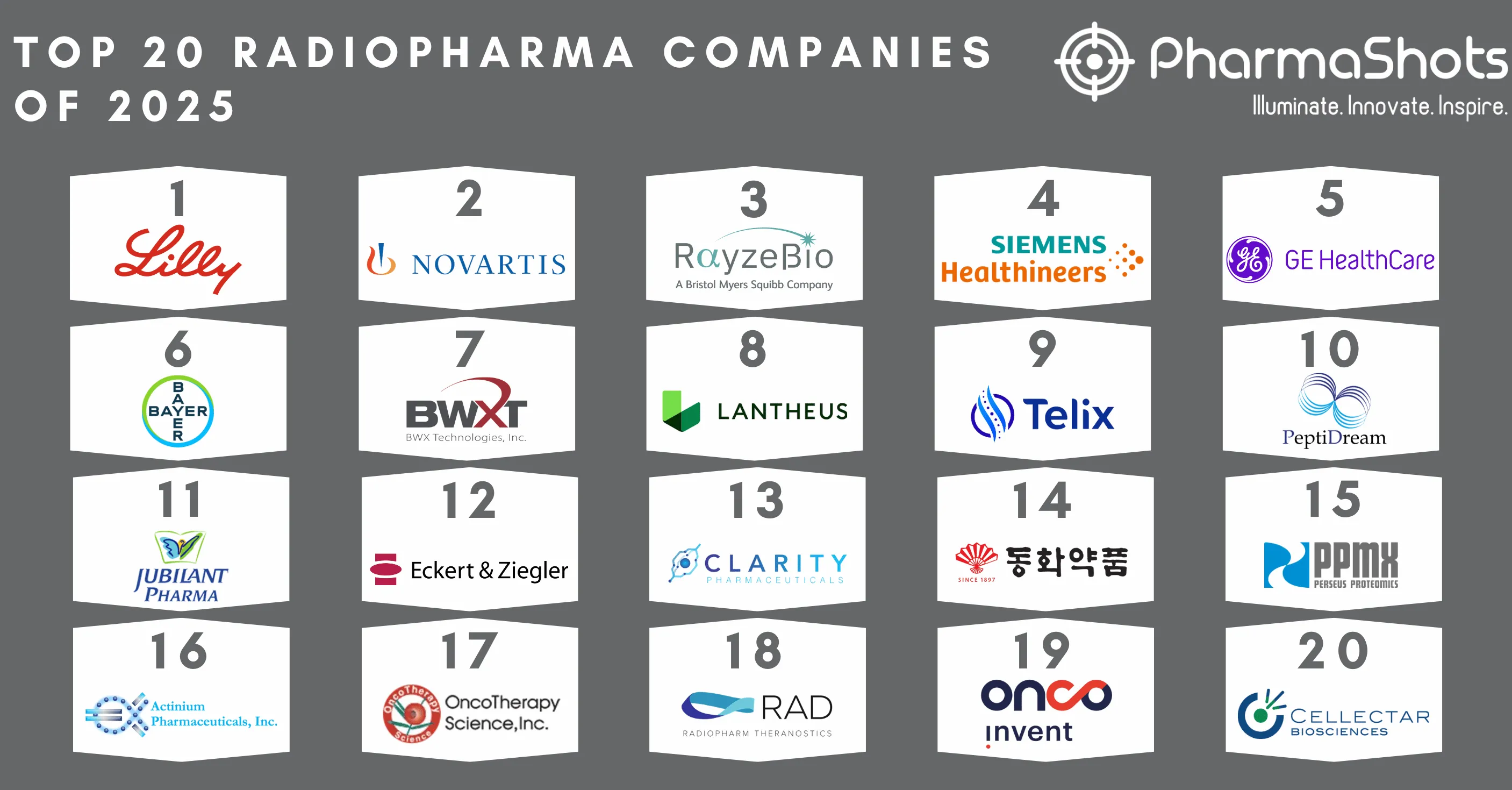
Top 20 Biopharma M&A of 2020 by Total Deal Value
- Biopharma merger and acquisition activity in 2020 was mainly filled with late-stage- bolt-on acquisitions- which were orders of magnitude smaller than the mega M&A deals of prior years.
- AstraZeneca holds the top rank by acquiring Alexion Pharmaceuticals among the top 20 acquisitions with a total deal value of $39B at a 1-day premium of 45% strengthening its capabilities in immunology- neurology- metabolic cardiovascular- nephrology- and rare disease.
- This article is based on the 2020 deals data as provided by the DealForma database. Our team at PharmaShots compiled a list of the top 20 acquisitions of 2020 based on total deal value.

Deal Date: Dec 17- 2020
Deal Value: $0.77B
Novartis acquired Cadent Therapeutics. The acquisition strengthens Novartis' neuroscience portfolio with the addition of Cadent's NMDAr program- including CAD-9303- an NMDAr positive allosteric modulator in Phase I for schizophrenia- and MIJ-821- an NMDAr negative allosteric modulator in Phase I for treatment-resistant depression. CAD-9303 and MIJ-821 were licensed to Novartis in 2015. Additionally- Novartis will gain full rights to CAD-1883- a clinical-stage SK channel positive allosteric modulator for movement disorders. Cadent Therapeutics received $210M up front and is eligible for up to $560M in milestones.

Deal Date: Aug 11- 2020
Deal Value: $0.87B
Bayer acquired KaNDy Therapeutics with its Phase IIb compound- NT-814- a small molecule for menopause and vasomotor symptoms. The acquisition strengthens Bayer's drug development portfolio in women's healthcare. KaNDy Therapeutics received $425M up front and is eligible for up to $450M in R&D milestones and additional undisclosed sales-based milestones.

Deal Date: Aug 25- 2020
Deal Value: $0.93B
ACADIA Pharmaceuticals acquired CerSci Therapeutics with its lead candidate- ACP-044- a Phase I small molecule for acute & chronic pain. The acquisition strengthens ACADIA's non-opioid pain therapies portfolio. CerSci shareholders received $52.5M up front and are eligible for up to $887M in development and sales-based milestones.

Deal Date: Dec 15- 2020
Deal Value: $1.04B
Eli Lilly acquired Prevail Therapeutics. The acquisition strengthens Eli Lilly's gene therapies portfolio with Prevail's clinical and preclinical disease-modifying AAV9 gene therapy assets in neuroscience. Prevail's pipeline includes PR001 in Phase I/II for Parkinson's disease and in preclinical studies for neuronopathic Gaucher disease- and PR006 in Phase I/II for frontotemporal dementia with GRN mutations. Prevail shareholders received $880M up front at a purchase price of $22.50 per share which represents a premium of approximately 117% plus 1 non-tradable CVR worth up to $4.00 per share in cash (or an aggregate of approximately $160M)- for a deal total of up to $26.50 per share in cash- approximately $1.04B. The CVR is payable upon the first regulatory approval for the commercial sale of a Prevail product in one of the countries- including the US- Japan- UK- Germany- France- Italy- or Spain.

Deal Date: Jan 10- 2020
Deal Value: $1.1B
Eli Lilly acquired Dermira with its IL-13 inhibitor antibody lebrikizumab- which is in Phase III for moderate-to-severe atopic dermatitis- and its marketed QBREXZA (glycopyrronium) medicated cloth for the topical treatment of primary axillary hyperhidrosis (uncontrolled excessive underarm sweating). Dermira licensed lebrikizumab from Genentech/Roche- which acquired the compound when it acquired Tanox- the originator. Dermira shareholders received $18.75 per share in cash at a 1-day premium of 2.2% for approximately $1.1B.

Deal Date: Oct 15- 2020
Deal Value: $1.3B
Lilly acquired Disarm Therapeutics. The acquisition strengthens Lilly's neurology portfolio with the addition of preclinical SARM1 inhibitors- a small molecule for peripheral neuropathy and other neurological diseases such as amyotrophic lateral sclerosis (ALS) and multiple sclerosis. Disarm shareholders received $135M up front and are eligible for up to $1.225B in development- regulatory- and commercial milestones if Lilly develops and commercializes new medicines based on Disarm's approach.

Deal Date: May 5- 2020
Deal Value: $1.3B
Alexion Pharmaceuticals acquired Portola Pharmaceuticals including its balance sheet and debt of approximately $215M. The acquisition expands Alexion's hematology- neurology- and critical care portfolio. Portola shareholders received $18 per share in cash at a premium of 130% for a total of $1.38B.

Deal Date: Dec 10- 2020
Deal Value: $1.4B
Boehringer Ingelheim acquired NBE-Therapeutics. The acquisition strengthens Boehringer Ingelheim's oncology pipeline with NBE-002- Phase I immunoconjugates for TNBC- lung- and ovarian cancers. NBE-Therapeutics is eligible to receive up to EUR 1.18B ($1.428B).

Deal Date: Dec 10- 2020
Deal Value: $1.7B
Gilead Sciences acquired MYR. The acquisition strengthens Gilead's infectious disease and liver disease portfolio with the addition of hepcludex- a lipoprotein approved for Hepatitis D in Europe and in Phase III for Hepatitis D- in Phase II for Hepatitis B- in Phase I for dyslipidemia- and in preclinical studies for NASH and primary biliary cholangitis. MYR shareholders received EUR 1.15B ($1.39B) up front and are eligible for up to EUR 300M ($363.7M) in milestones.

Deal Date: Nov 6- 2020
Deal Value: $1.8B
Novo Nordisk acquired Emisphere Technologies for $1.8B. The acquisition strengthens Novo Nordisk's biologics segment with the addition of Eligen SNAC drug delivery technology. Additionally- Novo Nordisk will buy out its Eligen SNAC royalty stream obligations with MHR Fund Management- Emisphere's largest shareholder. Novo Nordisk partnered with Emisphere in 2007 to develop oral biologics by applying Eligen SNAC. Emisphere shareholders received $7.82 per share in cash representing a 15% 1-day premium at approximately $1.35B. Additionally- Novo Nordisk will pay $450M to MHR Fund in connection with its royalty stream obligations.

Deal Date: June 11- 2020
Deal Value: $2.1B
Novo Nordisk acquired Corvidia Therapeutics. The acquisition strengthens Novo Nordisk's cardiovascular disease portfolio with Phase II ziltivekimab- an antibody for inflammatory cardiovascular risk in CKD patients. Corvidia received $725M up front and is eligible for up to $1.375B in regulatory and sales-based milestones.

Deal Date: Aug 31- 2020
Deal Value: $2.6B
Nestle acquired Aimmune Therapeutics. The acquisition strengthens Nestle's food allergy portfolio with Palforzia- an approved protein for peanut allergy. Aimmune Therapeutics received $2.6B at $34.50 per share at a 1-day premium of 174%. Nestle previously acquired 25% of the company starting with their 2016 partnership.

Deal Date: Nov 5- 2020
Deal Value: $2.7B
Merck acquired VelosBio. The acquisition strengthens Merck's oncology pipeline with the addition of Phase II VLS-101 antibody-drug conjugate targeting ROR1 for hematologic malignancies and solid tumors plus preclinical ADCs and bispecific antibodies. VelosBio shareholders will receive $2.75B.

Deal Date: Aug 17- 2020
Deal Value: $3.6B
Sanofi acquired Principia Biopharma. The acquisition strengthens Sanofi's autoimmune and allergic diseases portfolio with tolebrutinib (SAR442168)- a small molecule in Phase III for multiple sclerosis and in Phase II for CNS disorders. Principia Biopharma received $3.6B at a purchase price of $100 per share at a premium of 10%. In 2017- Sanofi and Principia partnered on the development and commercialization of SAR442168- which was in Phase IIb for multiple sclerosis. Principia's lead compound was rilzabrutinib- which was in Phase III for pemphigus.

Deal Date: Oct 26- 2020
Deal Value: $4B
Bayer acquired Asklepios BioPharmaceutical for $4B. The acquisition strengthens Bayer's cell and gene therapy portfolio with the addition of AskBio's AAV technology platform- CDMO facilities- and preclinical and clinical compounds for neuromuscular- CNS- cardiovascular- and metabolic diseases. Its leading gene therapy is in Phase I/II for Pompe disease. This also complements Bayer's 2019 acquisition of BlueRock Therapeutics. Asklepios is eligible to receive 75% of the milestone payments in the next five years.

Deal Date: Mar 2- 2020
Deal Value: $4.9B
Gilead acquired Forty-Seven with its lead product magrolimab- a monoclonal antibody targeting CD47 in Phase Ib for acute myeloid leukemia (AML). The company is also studying myelodysplastic syndrome (MDS) and diffuse large B-cell lymphoma (DLBCL). The acquisition strengthens Gilead's oncology portfolio. Forty-Seven received $4.9B at $95.50 per share at a premium of 65%.

Deal Date: Aug 19- 2020
Deal Value: $6.5B
Johnson & Johnson acquired Momenta Pharmaceuticals for $6.5B at $52.50 per share at a premium of 79%. The acquisition strengthens Johnson & Johnson's portfolio of autoimmune disease therapies with Phase III nipocalimab (M281)- an anti-FcRn antibody for warm autoimmune hemolytic anemia- which is also in Phase II for myasthenia gravis among other indications. Momenta's potential pipeline may have indications in maternal-fetal disorders- neuro-inflammatory disorders- rheumatology- dermatology- and autoimmune hematology. Nipocalimab recently received a rare pediatric disease designation.

Deal Date: Oct 05- 2020
Deal Value: $13.1B
Bristol Myers Squibb acquired MyoKardia for $13.1B at $225 per share at a 1-day premium of 61%. MyoKardia's mavacamten- a small molecule in Phase III for obstructive hypertrophic cardiomyopathy (HCM)- has an NDA submission expected in Q1 2021 and is in Phase II for Non-obstructive Hypertrophic Cardiomyopathy. MyoKardia also has 2 clinical compounds- danicamtiv (MYK-491) in Phase II for genetic dilated cardiomyopathy- systolic dysfunction- and atrial fibrillation- and MYK-224- a Phase I small molecule for hypertrophic cardiomyopathy.

Deal Date: Sep 13- 2020
Deal Value: $21B
Gilead acquired Immunomedics for $21B at $88.00 per share at a premium of 108%. The acquisition gives Gilead trodelvy- an antibody-drug conjugate approved for mTNBC. Trodelvy (sacituzumab govitecan-hziy) is a Trop-2 directed ADC- which received accelerated approval for mTNBC in the US. Trodelvy is also being studied in Phase III for 3L HR+/HER2- breast cancer and Phase II for bladder cancer- NSCLC- and other solid tumors.

Deal Date: Dec 12- 2020
Deal Value: $39B
AstraZeneca acquired Alexion for $39B including its drugs Soliris- Ultomiris- and 11 pipeline molecules in 20 different clinical programs. Soliris is an anti-complement component 5 mAb approved for paroxysmal nocturnal haemoglobinuria- atypical haemolytic uremic syndrome- generalized myasthenia gravis- and neuromyelitis optica spectrum disorder and is in Phase III for Guillain-Barre syndrome. Ultomiris is an approved antibody for Paroxysmal nocturnal hemoglobinuria and Hemolytic uremic syndrome and is in Phase III for generalized Myasthenia gravis- neuromyelitis optica spectrum disorder (NOSMD)- hematopoietic stem cell transplant-associated thrombotic microangiopathy- Amyotrophic lateral sclerosis- complement-mediated thrombotic microangiopathy- severe pneumonia or acute respiratory distress syndrome in COVID-19. Alexion's expertise in the complement cascade beyond C5 and additional modalities- including the pathway of the complement system- will strengthen AstraZeneca's portfolio including immunology- neurology- metabolic cardiovascular- nephrology- and rare disease. Alexion shareholders received $60 per share in cash and 2.1243 ADS shares of AstraZeneca valued at $175 per share at a premium of 45%- or approximately $39B. Upon completion- Alexion will own 15% of the combined company.
Related Post: Top 20 Generics Pharma Companies Based On 2019 Revenue
Tags

This content piece was prepared by our former Senior Editor. She had expertise in life science research and was an avid reader. For any query reach out to us at connect@pharmashots.com